Three Brazilian medical societies specializing in obesity and diabetes issued a joint statement criticizing the decision not to adopt weight loss pens containing liraglutide (Saxenda) and semaglutide (Ozempic and Wegovy) in the Unified Health System (SUS), a decision announced by the National Commission for the Incorporation of Technologies (Conitec) last Wednesday (20).
In the statement, the entities mention that there is an ‘elitization of access to obesity drug treatment in the country’ and that the decision not to offer the two medications in the public network ‘violates the beneficent principles of equity, universality, and integrality of the SUS.’
The note was signed by the Brazilian Association for the Study of Obesity and Metabolic Syndrome (Abeso), Brazilian Diabetes Society (SBD), and Brazilian Society of Endocrinology and Metabolism (SBEM). The three entities requested the incorporation of liraglutide into the SUS. The inclusion of semaglutide was requested by Novo Nordisk, the manufacturer of Ozempic and Wegovy. The Conitec’s denials were mainly justified by the high cost.
The medical societies point out that the country has six drugs for obesity treatment approved by the National Health Surveillance Agency (Anvisa), but inequality is a barrier to democratization. ‘Access to them is exclusive only for those who can afford it,’ the note states.
High Cost
The new Conitec decision maintains the opinion from May. At that time, the commission disclosed that including the two drugs in the public network could generate a minimum expenditure of R$3.4 billion over five years, reaching up to R$7 billion.
Challenging the cost argument, Abeso, SBD, and SBEM use the example of sibutramine, a low-cost obesity treatment drug that has not been evaluated by the SUS commission. ‘The decision (…), justified by the high cost of the medications, contrasts with the denial to even evaluate the incorporation of sibutramine, which has a monthly cost of less than 30 reais, an incorporation that was requested by these societies in December 2024.’
In the May decision, Conitec also highlighted the need for specialized monitoring, such as psychological support, for patients using the medication, making it difficult for widespread adoption in the public network. In June, after the opinion, a public consultation was opened to hear from civil society and healthcare professionals on the topic.
The three medical societies also emphasize that the lack of medications in the public system disproportionately affects vulnerable populations, for whom there is a lack of public policies to combat obesity. ‘Since 2019, Conitec has denied the incorporation of obesity drugs five times, always citing cost. Abeso, SBD, and SBEM disagree with the decision and lament for the patients, notably low-income black women living in peripheral areas, who have been most affected by obesity in recent years.’
Ultra-Processed Foods
The signatories of the statement also mention the lack of restrictions on access to ultra-processed foods, identified by scientists as one of the causes of the obesity epidemic worldwide.
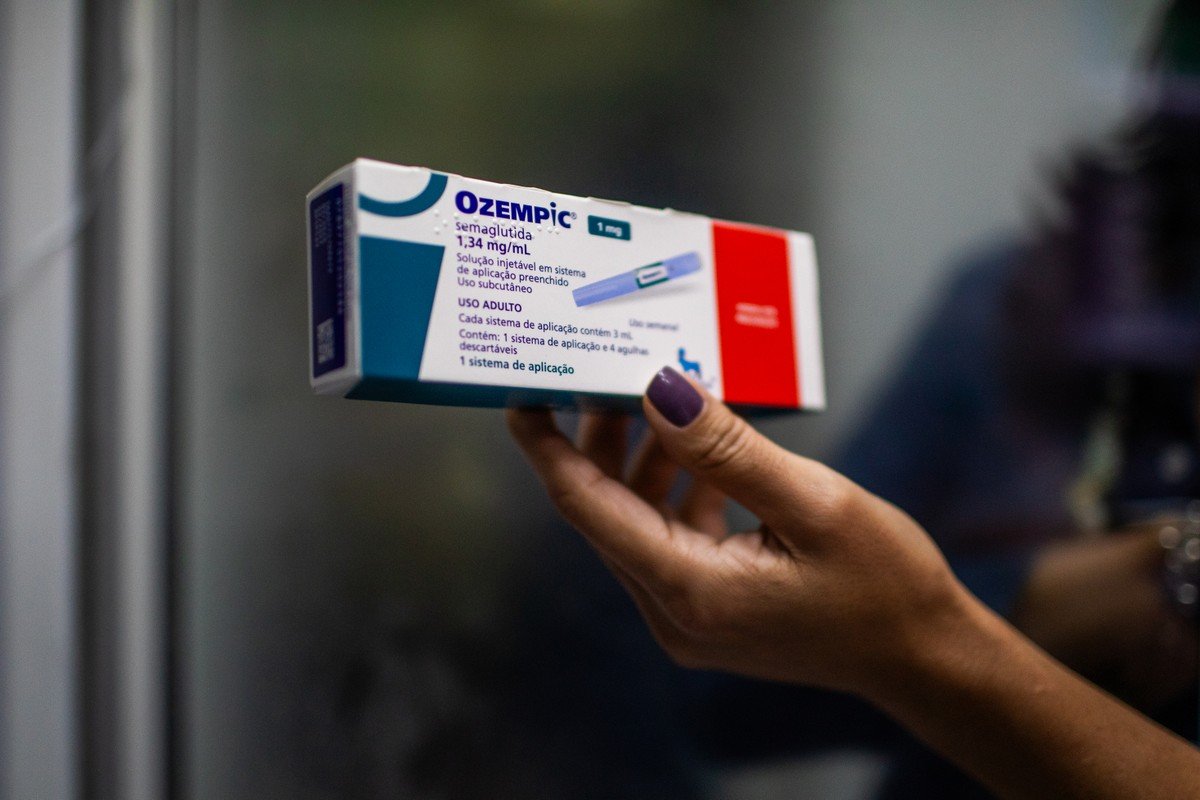
Medical Societies Criticize Decision Not to Include Ozempic and Saxenda in SUS: ‘Only for Those Who Can Afford It’

-

Flamengo and PSG have faced each other three times; check out their record
-

Indonesia Open Footgolf Tournament: Comedian Oki Rengga Admits Addiction, Wants to Become a Professional Athlete
-

Shameful Incident in Punjab! Landlord Rolls Tenant’s Daughter
-

Virgil van Dijk Expresses Desire for Mohamed Salah to Stay at Liverpool
Deixe um comentário